Latest News
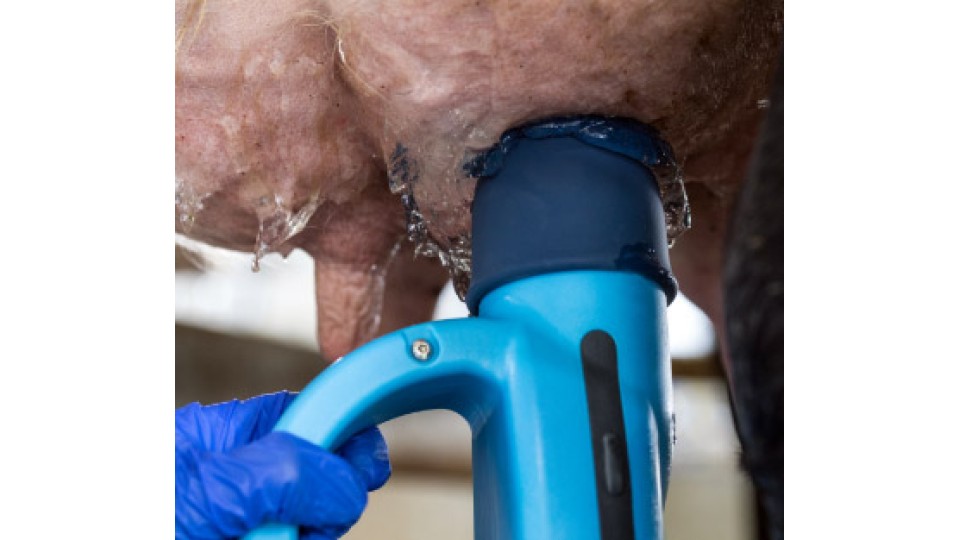
Why portable milkers can ease stress at calving
Why portable milkers can ease stress at calving- written by Shirley Macmillan and published in Far...
PRESS RELEASE on Acoustic Pulse Therapy and its application in mastitis.
PRESS NEWS
February 07, 2024
...
Scare the life out of mastitis with Double Barrel Dip!
“Double Barrel Dip” is a barrier dip for post-milking teat disinfection-
A 2-part mix that...
Udda-WOOL - UK wool for pre-milking teat preparation in dairy cows
UK wool can be a sustainable, value for money & effective teat wipe.
Pre-milking teat cleanin...
APT - Acoustic Pulse Technology for mastitis management in dairy cows
APT, or Acoustic Pulse Technology
WHAT IS ACOUSTIC PULSE TECHNOLOGY?
It is the local tran...
New anti-terrorism measures impact many farm chemicals.
Further to what was already in place to control access to chemicals like inorganic fertilisers, the ...
Extremely proud to WIN the Innovation Award at The Cream Awards
UK COMPANY TAKES NATIONAL AWARD FOR PLASTIC-SAVING AND CARBON-REDUCING SYSTEM FOR DAI...
Plasti-Save. Reducing plastic packaging and delivery miles in the dairy hygiene chemical supply chain.
Plasti-Save is a system to reduce plastic usage & delivery miles in the dairy hygiene chemical s...
Cutting carbon footprint begins with the calf (first published in British Dairying), written by Ann Hardy
The link between carbon footprint and efficiency is well recognised and most dairy farmers are now k...
Scour is the most common disease found in young calves, and the greatest cause of mortality.
Scour is the most common disease found in young calves, and the greatest cause of mortality, account...
BactoBAN dairy cleaning system shows good results, reducing bactoscan from over 50 to 14
Graham Whitting farms at Blackford Mill Farm, Redmarley, Gloucestershire. He milks 500 cows twice a ...